|
(those pairs of electrons shared between 2 atoms in the formation of a covalent bond)Ģ. (those electron pairs not used in the formation of covalent bonds) lone pairs (non-bonding pairs) of electrons.Remember that the pairs of dots in a Lewis structure (electron dot diagram) represent pairs of valence electrons (an 'electron cloud'). Electron pairs in the valence shell of an atom orient themselves so that their total energy is minimized.Įlectron pairs approach the nucleus of the atom as closely as possible while at the same time they stay away from each other as far as possible. Valence shell electron pair repulsion theory rests on 3 assumptions:ġ. Valence Shell Electron Pair Repulsion theory ( VSEPR theory, which is usually pronounced 'vesper'), also known as 'electron cloud' repulsion theory, is a method used to predict the shape of a molecule. No ads = no money for us = no free stuff for you! Valence Shell Electron Pair Repulsion (VSEPR) Theory Valence Shell Electron Pair Repulsion (VSEPR) theory is also referred to as 'Electron Cloud' Repulsion Theory.Valence Shell Electron Pair Repulsion (VSEPR) theory is used to predict shapes and bond angles of simple molecules.A diatomic molecule, a molecule composed of only 2 atoms, must always be linear in shape as the centres of the 2 atoms will always be in a straight line.The shape of a molecule, its geometry, is a description of the way the atoms in the molecule occupy space. 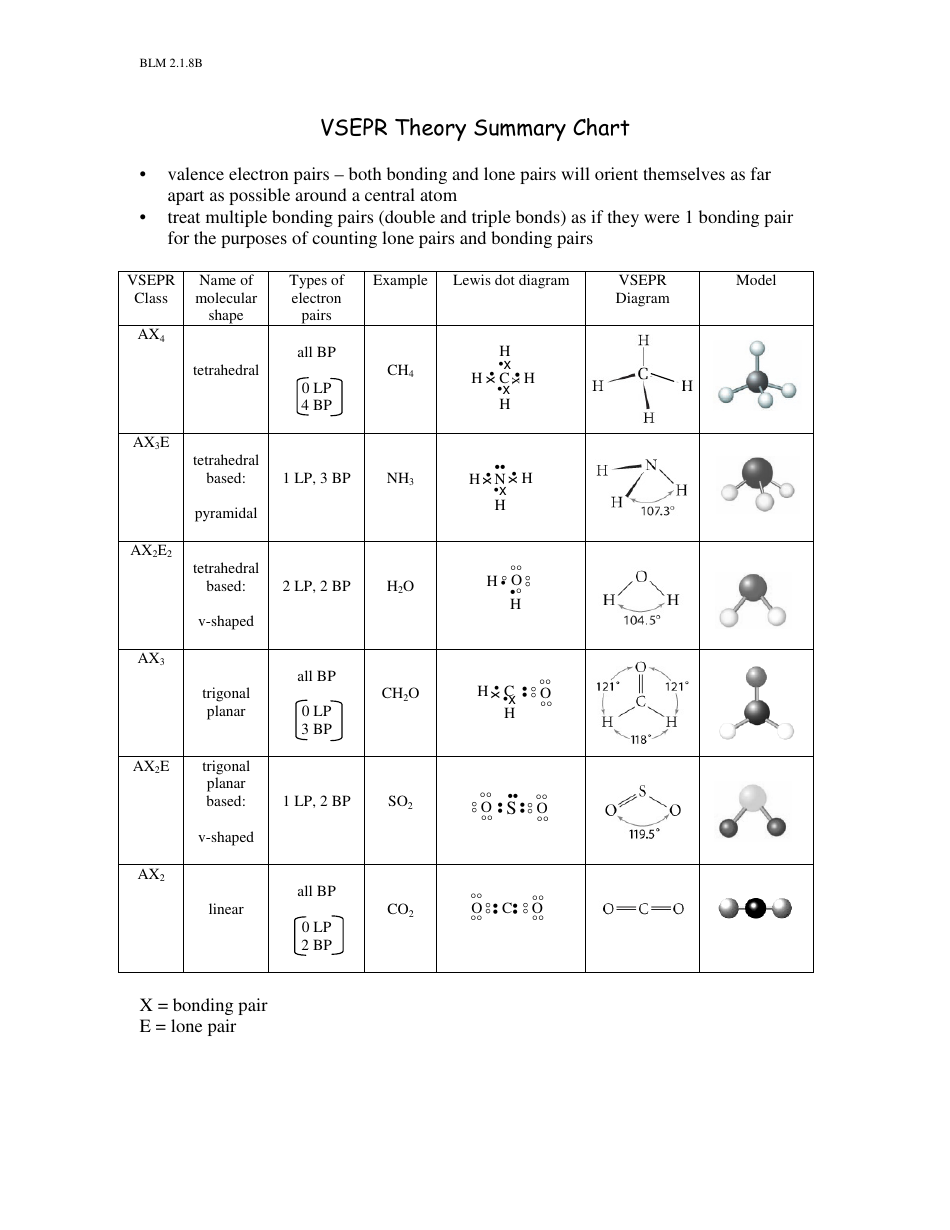
A covalent bond consists of 2 electrons shared between the 2 atoms (a bonding pair of electrons).A molecule consists of 2 or more atoms joined together by covalent bonds.You need to become an AUS-e-TUTE Member! Shapes of Molecules (VSEPR theory) Chemistry Tutorial Key Concepts Want chemistry games, drills, tests and more? Shapes of Molecules Chemistry Tutorial More Free Tutorials Become a Member Members Log‐in Contact Us
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
